Cystic Fibrosis External Quality Assessment process
The CF EQA Scheme is accredited by BELAC conform the ISO 17043 (PT-215), which is the international standard for conformity assessment of proficiency testing.
(PT-215), which is the international standard for conformity assessment of proficiency testing.
First time participants
Laboratories interested to participate in the CF EQA scheme need to have a username and password. With this username and password the laboratory can log in to the EQA participant's area. In the participants area the laboratory can access the registration form for the current EQA scheme, the laboratory can submit reports and consult results of previous years. In case your laboratory does not have a username and password, please get one by filling in this form.
Repeat participants
CF 2019 EQA timeline
January 2019: Distribution of the samples to the registered participants
February - March 2019: Datasheet open; submission of genotype results and written clinical reports
April 2019: Release of expected genotypes
May 2019: Assessment of the results
June 2019: General evaluation report and individual comments of the CF EQA scheme available online
Registration
All laboratories that have an account (username and password) will receive an invitation via e-mail to register for the CF EQA scheme online. An identification number (e.g. 20190000) is assigned to each participant automatically upon registration. Make sure you mention this identification number in all correspondence and on reports and raw data.
A copy of the registration form will be sent to the email address of all active contact persons linked to the laboratory. When the confirmation e-mail did not arrive, please check whether the final 'Submit' button was clicked on the registration form.
The CF network collaborates with the HGSA/RCPA Quality Assurance Programs (QAP) for the registration of Australasian laboratories. These laboratories should enroll through the HGSA/RCPA QAP.
Payment information
All participating laboratories should pay a registration fee of 275 EUR. When a registration form is submitted, we assume that your laboratory participates in the scheme. And thus a registration fee has to be paid, independently whether you submit results or not.
An invoice will be generated and sent to the laboratory by e-mail once the registration form is submitted. This is a manual process which takes on average 15 work days. In addition, this invoice is attached to the EQA participant's area. For the Australasian laboratories, payment will be handled through the HGSA/RCPA QAP.
Please remit payment within 30 days after receipt of your invoice. Make sure you specify the correct structured communication (+++xxx/xxxx/xxxxx+++) with your payment. Without this reference, your payment cannot be processed automatically, which can cause a delay in the registration of the payment by our financial department, and other problems.
Payment can be made by bank transfer and without charges to the beneficiary:
Account holder: K.U.Leuven
Account number: 432-0000011-57
Bank's name: K.B.C.
Bank address: Bedrijvenkantoor Leuven, Brusselsesteenweg 100, B-3000 Leuven, Belgium
IBAN code: BE09 4320 0000 1157
BIC/SWIFT code: KREDBE BB
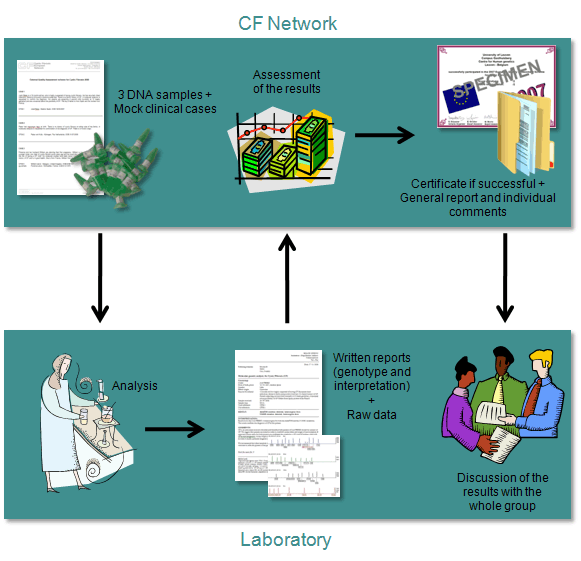
Samples
Three purified DNA samples (15µl, conc ~ 1µg/3µl), harboring homozygous or heterozygous CFTR mutations, will be sent to the registered laboratories with the request to test for the presence of CFTR mutations, using the routine protocols. The samples are accompanied by mock clinical cases.
New samples:
In case the CF EQA samples did not arrive or arrived broken, the package will be reshipped at our expense.
Additional samples:
The samples contain enough DNA to perform CFTR testing with the current available testing methods. However, if you need additional DNA, we can send additional samples, your laboratory will be charged 50 euro for the transport costs.
Analysis and reporting
The laboratories have about 8 weeks to analyze the samples with the detection assay they routinely use and should submit written reports and raw data. These reports and raw data should be uploaded via an electronic datasheet, available when the laboratory logs in to the CF Network website. The CF Network strongly advises NOT to send reports and raw data by postal mail or e-mail.
Report:
The laboratory should submit a report for each mock clinical case. A report represents the report a laboratory normally issues to the medical doctor who requested the CF test. Therefore genotype and interpretation information should be present in the reports submitted to the CF Network. Reporting is allowed in six languages: English, French, German, Spanish, Italian and Dutch. A report in the native language can be submitted in addition. This will be consulted when there is doubt that these errors are due to a translation problem. Laboratories may write anonymous reports, but this is not obligated, as all quality assessors signed a confidential agreement on this. Reports are preferable submitted as .doc, .docx or .pdf files. Example reports are available on the CF Network website. When using example reports, it is essential to formulate an interpretation based on the context of the clinical situation.
Raw data:
Raw data of the analysis (e.g. agarose gel, INNO-LiPA strip, ...) might be helpful during the assessment in case of unclear results. Raw data are preferable submitted as .tiff, .ppt, .pptx, .pdf, .jpg or .gif
Assessment
The scheme organizer together with a panel of quality assessors will evaluate the reports and raw data. A general evaluation report and individual comments are prepared and made available online. This is notified by e-mail to all active contact persons. A certificate of participation will be made available online for all participating laboratories. The certificate will specify whether participation was successful or not. Results of the CF EQA scheme will be sent only to those laboratories that paid the registration fee. Laboratories that did not participate successfully in the current year and at least once in the two previous years will receive an additional letter inviting them to contact the assessors to discuss their results and ways to improve their performance.
Appeals
The CF Network allows people to react on the score and/or individual comments they received. All appeals should be sent by e-mail to before mid-July. The CF Network will collect all appeals and the appeals will be discussed by the assessors. The CF Network will respond after August 29, then marks become final.
Last changed: 2019-12-04

 Contact
Contact Home
Home